The team describes the use of MiVO in combination with 3D human intestinal tissues for highly reliable in vitro intestinal permeation tests
Intestinal permeability is crucial in regulating the bioavailability and the biological effects of drugs and compounds. However, systematic and quantitative studies of absorption of molecules are quite limited due to the lack of reliable experimental models able to mimic in vivo responses. A recent study published in Altex sought to answer some of these questions.
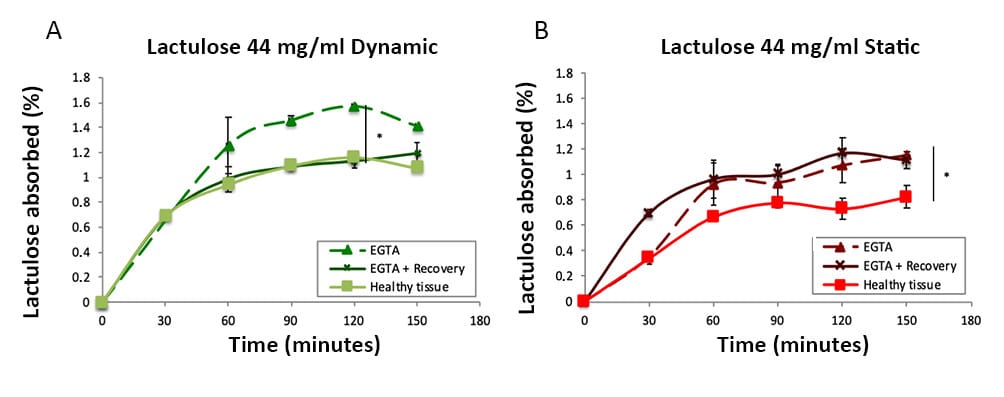
They adopted the fluidic MIVO® device for hosting and culturing 3D reconstructed intestinal epithelium, resembling in vitro the human fluid-dynamic intestinal environment. The platform has been investigated, both in healthy and induced pathological conditions, by monitoring the absorption of two non-metabolized sugars, lactulose and mannitol, frequently used as indicators of intestinal barrier dysfunctions. The results showed that an in vivo-like plateau of the percentage of absorbed sugars was reached for lactulose and mannitol and that the mannitol absorbed was much greater than the lactulose, in line with clinical data, thus revealing the good reliability of the model. Interestingly, after intestine damage treatment, the pattern of sugar passage comparable with the healthy tissue was completely restored only in dynamic conditions within MIVO® chamber, due to the beneficial role of the dynamic fluid flow beneath the membrane. Therefore, the MIVO® combined with EpiIntestinalTM™ platform can represent a reliable in vitro model of healthy and pathological small intestinal barrier to study the passage of molecules, by discriminating the two main intestinal absorption mechanisms.
A. Marrella, P. Buratti, J. Markus , G. Firpo, M. Pesenti , T. Landry , S. Ayehunie , S. Scaglione , H. Kandarova and M. Aiello (2019): “In vitro demonstration of intestinal absorption mechanisms of different sugars using 3D organotypic tissues in a fluidic device”. ALTEX – Alternatives to animal experimentation.
All Resources
Never stop learning!
Check publications from the team, protocols, and useful information to boost your research and get into organ on chip technology!